Viral Inactivation Market Size to Reach a Valuation of USD 2.27 Billion by 2035; Industry Growth is Driven by Increasing Demand for Virus Safety in Monoclonal Antibodies’ Production Process Globally | SNS Insider
U.S. Viral Inactivation Market was valued at USD 0.27 billion in 2025 and is expected to reach around USD 0.83 billion by 2035 owing to the its highly advanced manufacturing process
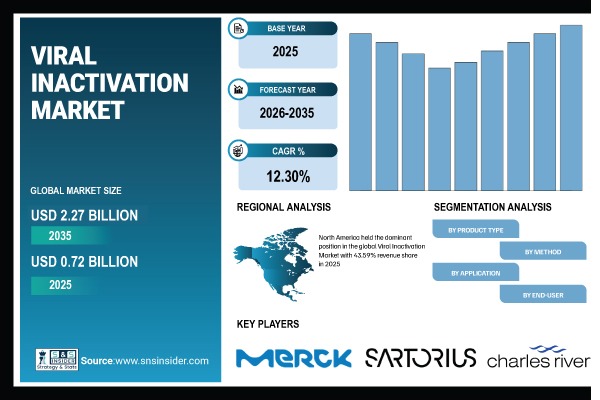
Austin, May 15, 2026 (GLOBE NEWSWIRE) — According to SNS Insider, The Viral Inactivation Market was valued at USD 0.72 Billion in 2025 and is expected to reach USD 2.27 Billion by 2035, growing at a CAGR of 12.30% from 2026–2035.
The market for viral inactivation is seeing considerable growth due to the rapid expansion of biologics and biosimilars production processes, increasing need for virus safety during the production process of monoclonal antibodies and recombinant proteins, and increasing outsourcing of bioprocessing services by CMOs/CDMOs.
Get Exclusive Access to the Sample Report of Viral Inactivation Market Forecast @ https://www.snsinsider.com/sample-request/10170
The U.S. Viral Inactivation Market was valued at USD 0.27 Billion in 2025 and is expected to reach around USD 0.83 Billion by 2035, growing at a CAGR of 11.88% from 2026–2035.
The USA has the maximum market share in comparison to all other countries in the world due to the presence of a highly developed manufacturing process, biologic drug makers, and CDMOs. The rise in the utilization of monoclonal antibodies, recombinant proteins, vaccines, and cell and gene therapies has increased the requirement for viral inactivation methods.
The Europe Viral Inactivation Market is estimated to be USD 0.23 Billion in 2025 and is projected to reach USD 0.69 Billion by 2035, growing at a CAGR of 11.67% during 2026–2035.
In 2025, Europe comprised a significant share in the international market owing to its elaborate research framework in terms of biologics, advanced manufacturing capabilities in the pharmaceuticals industry, and favorable regulations promoting advanced therapeutics.
Rising Demand for Biologics, Vaccines, and Advanced Therapies Propel Market Growth Globally
Viral Inactivation Market Key Structural Factors can be linked to the increasing tendency of biologics production, which comprises such substances as monoclonal antibodies, recombinant proteins, vaccines, and plasma products, which have to be produced without viruses because it is mandatory according to law. Due to the growing diversity and complexity of the biologics pipeline, companies tend to use viral inactivation technologies, which comprise solvents/detergents process, low pH process, and nanofiltration.
Segmentation Analysis:
By Product Type
In 2025, the Reagents & Kits segment maintained its dominant position in the Viral Inactivation Market, accounting for 41.36% share attributed to their widespread use across biopharmaceutical manufacturing workflows. From 2026 to 2035, the Pathogen Reduction Systems segment is projected to record the highest CAGR driven by increasing demand for advanced and multi-modal viral safety solutions globally.
By Method
The Solvent/Detergent (S/D) Inactivation segment held the largest share of 44.15% in 2025 propelled by its longstanding regulatory approval and proven efficacy against enveloped viruses. The Low pH Inactivation segment is expected to register the highest CAGR during the 2026–2035 forecast period supported by increasing adoption in monoclonal antibody purification processes globally.
By Application
In 2025, the Monoclonal Antibody Production segment maintained its dominant position in the Viral Inactivation Market, accounting for 33.85% share fueled by the huge global demand for therapeutic antibodies for oncology, autoimmune diseases, and infections. From 2026 to 2035, cell and advanced biologics-related applications (included under Others) are projected to record the highest CAGR fueled by the rapid expansion of cell therapy, gene therapy, and regenerative medicine pipelines globally.
By End-User
The Pharmaceutical & Biopharmaceutical Companies segment maintained the highest end-user share of 46.24% in 2025 due to the strength of their internal capacity to manufacture biological drugs globally. The Contract Manufacturing Organizations (CMOs/CDMOs) segment is projected to achieve the highest growth rate during 2026–2035.
By Scale of Operation
The Preclinical & R&D Scale segment held the largest share of 32.51% in 2025 due to the widespread application of viral inactivation procedures at the early stages of biologics development globally. The Commercial Manufacturing Scale segment is projected to register the highest CAGR during 2026–2035.
Get Expert-Led Insights for Your Business Strategy — Connect with Analysts Now @ https://www.snsinsider.com/request-analyst/10170
Regional Insights:
The North American region was the leading player in the worldwide Viral Inactivation Market, accounting for a revenue market share of 43.59% in 2025, due to its advanced biologics manufacturing environment, stringent compliance standards, and early adoption of the viral safety process validation technique.
The highest CAGR of 13.33% during the forecast period (2026-2035) will be recorded in the Asia Pacific market due to the fast-paced growth in biologics production capacity, high demand for outsourced manufacturing services, and favorable government initiatives towards biotechnology.
Major Players Analysis Listed in the Viral Inactivation Market Report:
- Merck KGaA
- Sartorius AG
- Charles River Laboratories
- Thermo Fisher Scientific Inc.
- Lonza Group
- Cytiva (Danaher Corporation)
- WuXi AppTec
- Pall Corporation (Danaher)
- Texcell SA
- Eurofins Scientific
- SGS SA
- Cerus Corporation
- Terumo Blood and Cell Technologies
- Rad Source Technologies Inc.
- Bio-Rad Laboratories
- Parker Hannifin Corporation
- Catalent Inc.
- Boehringer Ingelheim BioXcellence
- Clean Cells (Sartorius Group)
- Syngene International Ltd.
Recent Developments:
- 2026: Lonza Group expanded its global biologics manufacturing network with enhanced viral safety integration across new large-scale CDMO facilities in the U.S. and Europe, incorporating advanced solvent/detergent and nanofiltration systems to support rising demand for monoclonal antibody and cell therapy production.
- 2026: Thermo Fisher Scientific strengthened its bioprocessing portfolio by upgrading its viral safety workflow solutions, including improved single-use filtration systems and automated viral clearance validation tools, supporting faster scale-up of biologics manufacturing in North America and Asia-Pacific.
Purchase Single User PDF of Global Viral Inactivation Market Intelligence Report @ https://www.snsinsider.com/checkout/10170
Exclusive Sections of the Report (The USPs):
- MARKET ADOPTION & UTILIZATION DYNAMICS METRICS – helps you understand adoption rates of viral inactivation workflows across biologics and vaccine manufacturing, usage trends of solvent/detergent methods, integration with downstream purification processes, and adoption growth in CMOs/CDMOs versus in-house production facilities.
- PROCESS EFFICIENCY & BIO-SAFETY PERFORMANCE METRICS – helps you evaluate viral clearance efficiency, contamination risk reduction, batch release success rates, biologics recovery efficiency after inactivation, and variability in viral safety performance across regional GMP standards.
- TECHNOLOGICAL ADVANCEMENT & INNOVATION METRICS – helps you analyze adoption of next-generation viral inactivation technologies such as nanofiltration, UV-based systems, AI-monitored platforms, automated viral safety systems, and R&D intensity focused on improving yield retention and product safety.
- INDUSTRY DEMAND, REGULATION & COMMERCIAL DYNAMICS METRICS – helps you assess the impact of biologics production expansion, regulatory dependency across FDA/EMA/PMDA frameworks, outsourcing trends to CDMOs, accessibility of standardized viral inactivation kits, and growth contribution from emerging biopharmaceutical markets.
- BIOLOGICS SAFETY & COMPLIANCE METRICS – helps you identify trends in viral safety validation, regulatory compliance effectiveness, contamination prevention strategies, and quality assurance standards across biologics, plasma-derived products, and vaccine manufacturing pipelines.
Viral Inactivation Market Report Scope
| Report Attributes | Details |
| Market Size in 2025 | USD 0.72 Billion |
| Market Size by 2035 | USD 2.27 Billion |
| CAGR | CAGR of 12.30% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Key Segments | • By Product Type (Reagents & Kits, Detergents (Solvent/Detergent Systems), pH Adjustment Solutions, Pathogen Reduction Systems, Viral Filtration Systems, Other Consumables) • By Method (Solvent/Detergent (S/D) Inactivation, Heat Treatment (Pasteurization), Low pH Inactivation, UV-C Irradiation, Nanofiltration-Based Inactivation, Others) • By Application (Monoclonal Antibody Production, Vaccines, Blood & Plasma Derivatives, Recombinant Proteins, Cell & Gene Therapy Products, Stem Cell Research, Others) • By End-User (Pharmaceutical & Biopharmaceutical Companies, Contract Manufacturing Organizations (CMOs/CDMOs), Research Institutes & Academic Laboratories, Blood Banks & Plasma Fractionation Centers, Biotechnology Companies) • By Scale of Operation (Preclinical & R&D Scale, Clinical Scale, Commercial Manufacturing Scale) |
| Regional Analysis/Coverage | North America (US, Canada), Europe (Germany, UK, France, Italy, Spain, Russia, Poland, Rest of Europe), Asia Pacific (China, India, Japan, South Korea, Australia, ASEAN Countries, Rest of Asia Pacific), Middle East & Africa (UAE, Saudi Arabia, Qatar, South Africa, Rest of Middle East & Africa), Latin America (Brazil, Argentina, Mexico, Colombia, Rest of Latin America). |
Rising Demand for Viral Inactivation Market Data, Our Full Report Trend Analysis @ https://www.snsinsider.com/reports/viral-inactivation-market-10170
Other Trending Related Report:
Sterilization Equipment Market
Medical Device Sterilization Market
Cell & Gene Therapy Manufacturing Services Market
Cell and Gene Therapy CDMO Market
About Us:
SNS Insider is one of the leading market research and consulting agencies that dominates the market research industry globally. Our company’s aim is to give clients the knowledge they require in order to function in changing circumstances. In order to give you current, accurate market data, consumer insights, and opinions so that you can make decisions with confidence, we employ a variety of techniques, including surveys, video talks, and focus groups around the world.
CONTACT: Contact Data Contact Us: Rohan Jadhav - Principal Consultant Phone: +1-315 636 4242 (US) | +44- 20 3290 5010 (UK)
 Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. DailyIndiaNews.com takes no editorial responsibility for the same.
Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. DailyIndiaNews.com takes no editorial responsibility for the same.